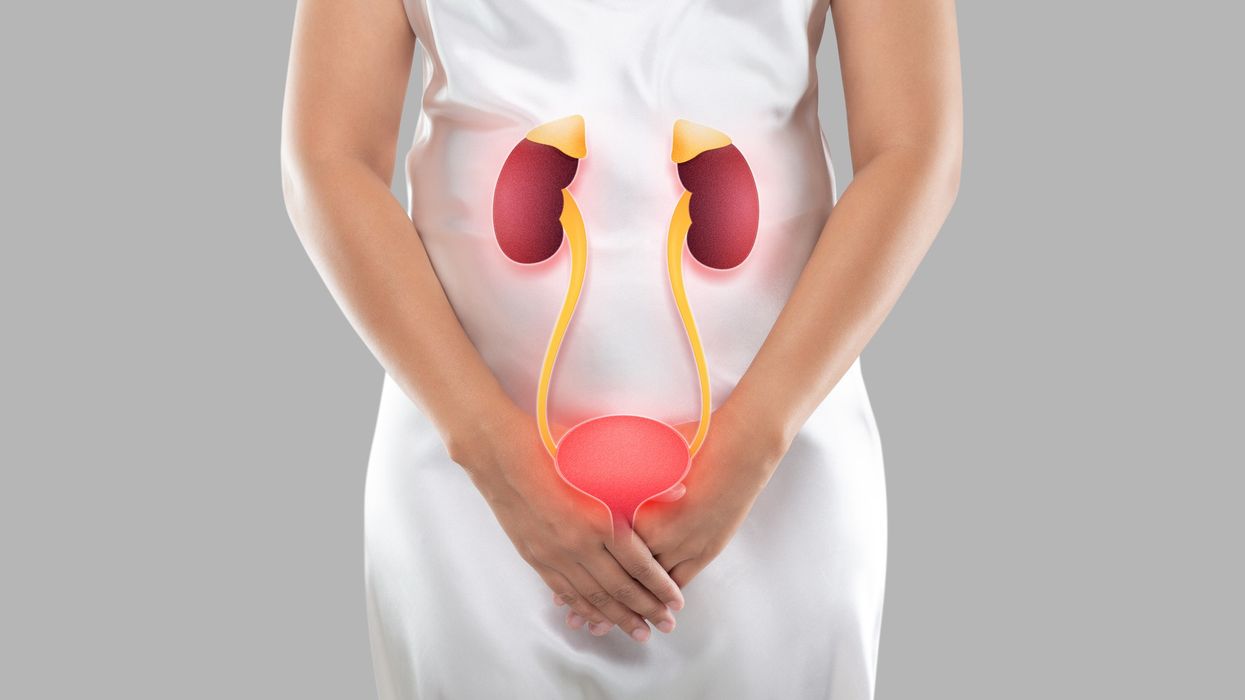
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved an oral antibiotic pill to treat uncomplicated urinary tract infections (UTIs) for women for the first time in three decades.
In the antibiotic, gepotidacin (also known as Blujepa), the active ingredient targets and blocks two enzymes that bacterias need to replicate and multiply, making it effective against many drug-resistant infections such as E. coli.
The recommended dose is two tablets twice daily for five days, approximately every 12 hours, and it can be used by females over the age of 12 and weigh at least 40kg.
A UTI is a bladder infection, usually caused by bacteria like E.coli, and they are much more common in women than men.
Uncomplicated UTIs are the most common bacterial infection, affecting around half of women in the UK.
With drug-resistant bacteria increasing, new treatment options are critical in preventing treatment failure and complications, including sepsis or permanent kidney damage.
The MHRA’s national approval is supported by evidence from two multicentre, randomised, active-controlled Phase 3 trials in females with uncomplicated UTIs.
In both trials, gepotidacin was shown to be as effective as nitrofurantoin.
The most common side effects of the medicine (which may affect more than 1 in 10 people) include diarrhoea and nausea, both of which were considered mild.
MHRA Interim Executive Director Julian Beach said, “As the first new type of oral antibiotic to treat uncomplicated UTIs to be approved in nearly three decades, gepotidacin provides a new treatment option for women facing urinary tract infections that can severely impact daily life."