Clinicians have been instructed to avoid prescribing GLP-1 receptor agonists for off-label indications
The Department of Health and Social Care (DHSC) has issued an alert to update healthcare professionals on the shortage of glucagon-like peptide-1 receptor agonists (GLP-1 RAs), which are used to treat type 2 diabetes.
It has informed that the supply of the drugs continues to be limited, and is not expected to return to normal until at least the end of 2024.
An increase in demand for these products for licensed and off-label indications is cited as the cause of the supply issues.
“Rybelsus® (semaglutide) tablets are now available in sufficient quantities to support initiation of GLP1 RA treatment in people with type 2 diabetes (T2DM) in whom new initiation of GLP-1 RA therapy would be clinically appropriate,” the DHSC’s National Patient Safety Alert reads.
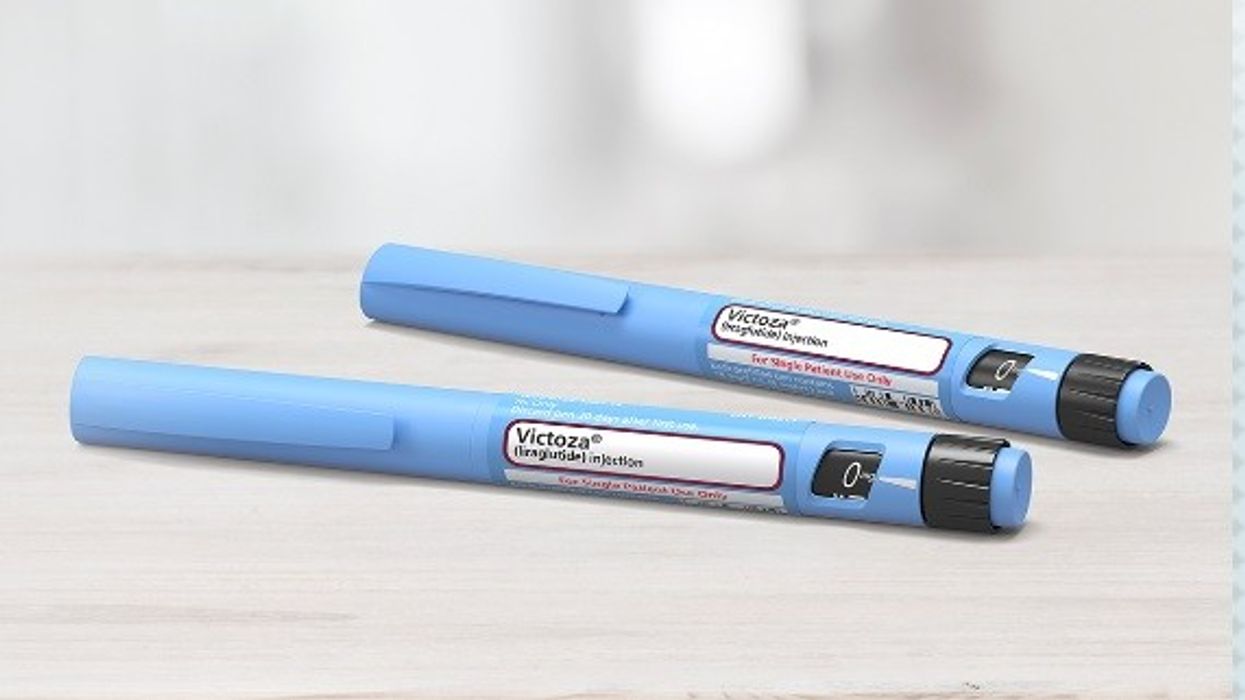
While Victoza® (liraglutide) continues to be out of stock, Byetta® (exenatide) 5micrograms/0.02ml and 10micrograms/0.04ml solution for injection 1.2ml pre-filled pens is to be discontinued from March 2024.
However, Saxenda® (liraglutide) and Wegovy® (semaglutide) are still available on the NHS via specialist weight management services.
Clinicians have been instructed to only prescribe GLP-1 RAs for licensed indications, and avoid prescribing for off-label indications.
“Existing stock must be conserved for patients with T2DM to mitigate the risk of impaired access to treatment and increased risk in diabetes related complications,” DHSC said.
The department has also raised concern that the absence of GLP-1 RA stock may cause surge in demand for insulin, and affect its supplies, which currently remain good.
Until supply issues have resolved, clinicians are advised to prescribe Rybelsus® tablets for new initiations of a GLP-1 RA, as well as for patients using Byetta® and Victoza® injections.
Prescribers are required to counsel their patients on any changes in drug, formulation, and dose regimen where appropriate.