Key Summary
- By the end of 2026, core patents on semaglutide will have expired in 10 countries, including Brazil, China, South Africa, Turkey and Canada.
- However, in the US, UK, and many parts of Europe, the patent protection for semaglutide lasts till 2031 and beyond.
- Dr Reddy's Laboratories is aiming to launch its version of semaglutide in Canada by May 2026
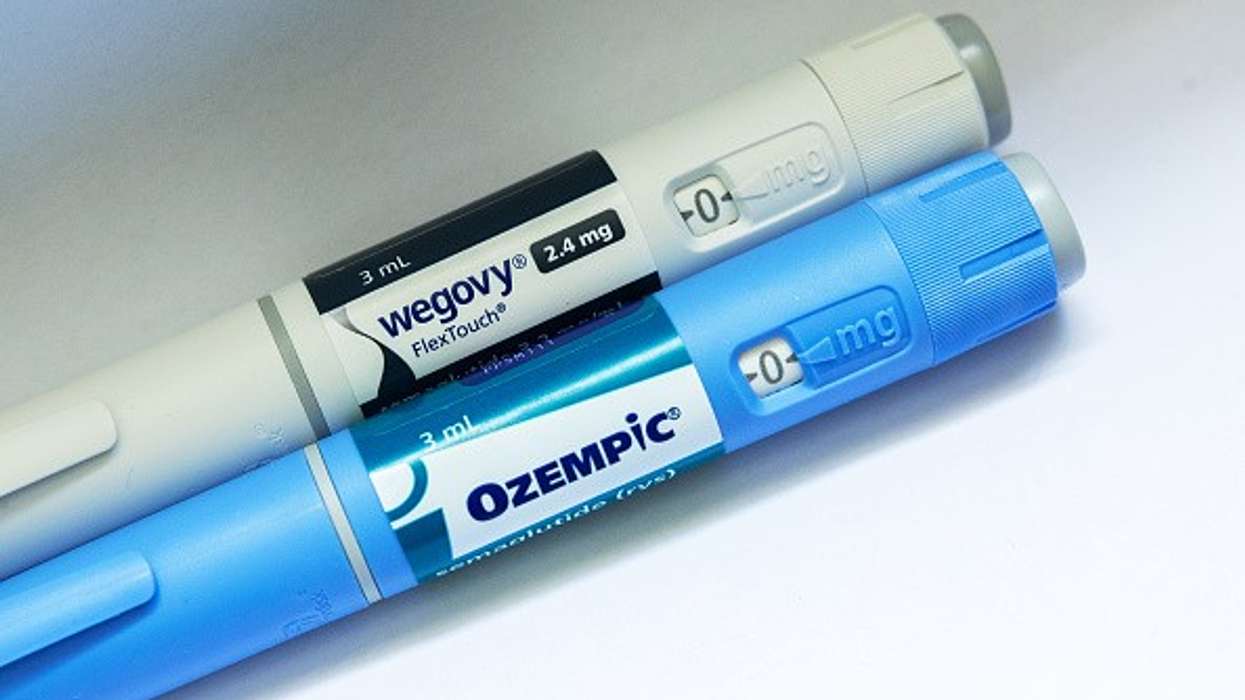
The expiry of semaglutide patent, the molecule in Novo Nordisk's weight-loss drugs Wegovy and Ozempic, in India is expected to shake up the country's drug industry, home to many generics majors.
Several Indian companies have firmed up plans to launch low-cost versions of the popular drug used to treat Type 2 diabetes and weight loss.
Sun Pharmaceutical Industries, Dr Reddy's Laboratories, Zydus Lifesciences, Lupin, and Mankind Pharma are expected to launch their generics version of semaglutide on Saturday (21), a day after the patent expires.
Sun Pharma has got approval for two branded options, Sematrinity for type‑2 diabetes and Noveltreat for chronic weight management.
Dr Reddy's Laboratories has obtained regulatory clearance for its semaglutide brand Obeda, while Mankind Pharma is launching Samakind.
Zydus will market a 15 mg reusable pen under the brand names Semaglyn, Mashema and Altreme, while Lupin will co-market Zydus’ injection in India under the brand names Semanext and Lupin’s Livarise.
Price drop
The prices are expected to fall by more than half and will boost access for people in India, and eventually in other countries too.
By the end of 2026, core patents on semaglutide will have expired in 10 countries that represent 48 percent of the global obesity burden, according to a study published earlier this month by researchers.
These include Brazil, China, South Africa, Turkey and Canada.
However, in the US, UK, and many parts of Europe, the patent protection for semaglutide lasts till 2031 and beyond.
Research firm Pharmarack estimates the Indian market will soon be flooded with options.
"What we understand is, there will be more than 50 brands that will be launched in the market and there are more than 40 players who will be launching these drugs," Pharmarack's vice president Sheetal Sapale told AFP.
In India, Wegovy and Ozempic were confined to affluent patients, as most Indians pay for healthcare out of own pocket.
But the impending price drop is expected to substantially broaden its user base.
The entry of generics will also challenge Eli Lilly, India's top-selling drug by value, according to Pharmarack.
India’s weight loss drug sales have grown 10-fold in five years to $153 million as of 2026, and are projected to soar to over half a billion by 2030.
Investment bank Jefferies predicts that India's semaglutide market could eventually reach $1bn domestically with the right pricing and uptake.
Ripple effect in other countries
The global impact may prove even more profound. India supplies more than half of Africa's generic medicines, and cheaper semaglutide could become a lifeline for countries where obesity is rising rapidly, but treatment remains unaffordable.
"Lower‑cost semaglutide could significantly expand access to effective treatment, particularly in middle-income countries where price has been a major barrier," Simon Barquera, president of the World Obesity Federation, told AFP.
"But medication alone will not reverse the global rise in obesity. Obesity is a complex, chronic disease," he said, noting the importance of prevention efforts and healthier environments.
Indian firms will be a key driving force, with Dr Reddy's Laboratories aiming to launch its version of semaglutide in Canada by May 2026.
Rising obesity
While the country still accounts for a third of the world's undernutrition, according to the World Health Organization (WHO), rising incomes and urban lifestyles have sharply increased obesity rates.
Government data released in March last year shows 24 percent of women and 23 percent of men are overweight or obese in India.
India could have over 440 million overweight or obese people by 2050, a study published in The Lancet showed.
It also has the most adults suffering from diabetes after China, according to the International Diabetes Federation.
Side effects
But using such drugs can cause side effects, including nausea and gastrointestinal issues.
The Medicines and Healthcare products Regulatory Agency (MHRA) recently revised the product information for semaglutide, noting a rare risk of vision loss.
It had earlier updated the product information to include the small risk of severe acute pancreatitis in patients taking GLP-1s.
GLP-1s or Glucagon-Like Peptide-1 receptor agonists include semaglutide (Wegovy, Ozempic) and tirzepatide (Mounjaro).