Key Summary
- The MHRA has approved Deuruxolitinib (Leqselvi) to treat adults with severe Alopecia Areata.
- The drug works by blocking inflammation-related enzymes and helping hair regrow, based on trials involving 1,223 patients.
- It is prescription-only, taken 8 mg twice daily, with possible side effects including headache and acne.
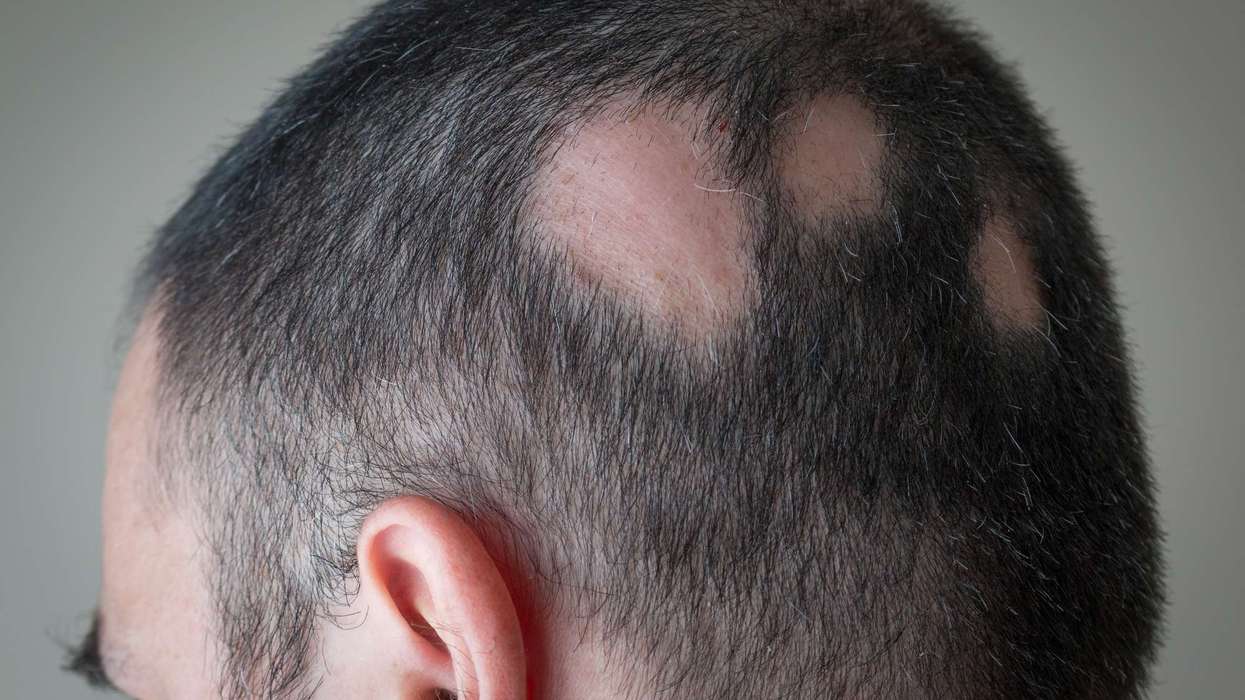
The Medicines and Healthcare products Regulatory Agency (MHRA) approved deuruxolitinib (Leqselvi) to treat adult patients with severe alopecia areata, an auto immune condition that leads to inflammation of hair follicles.
This inflammation results in hair loss on scalp, face and other body parts.
Deuruxolitinib (Leqselvi) was proven as an effective remedy after two pivotal clinical trials that involved 1,223 adult patients.
The drug controls the enzyme activity of JAK1, JAK2 AND TYK2 relative JAK3 kinases causing the inflammation.
The consumption of the tablet will calm the inflammation and promotes the regrowth of hair.
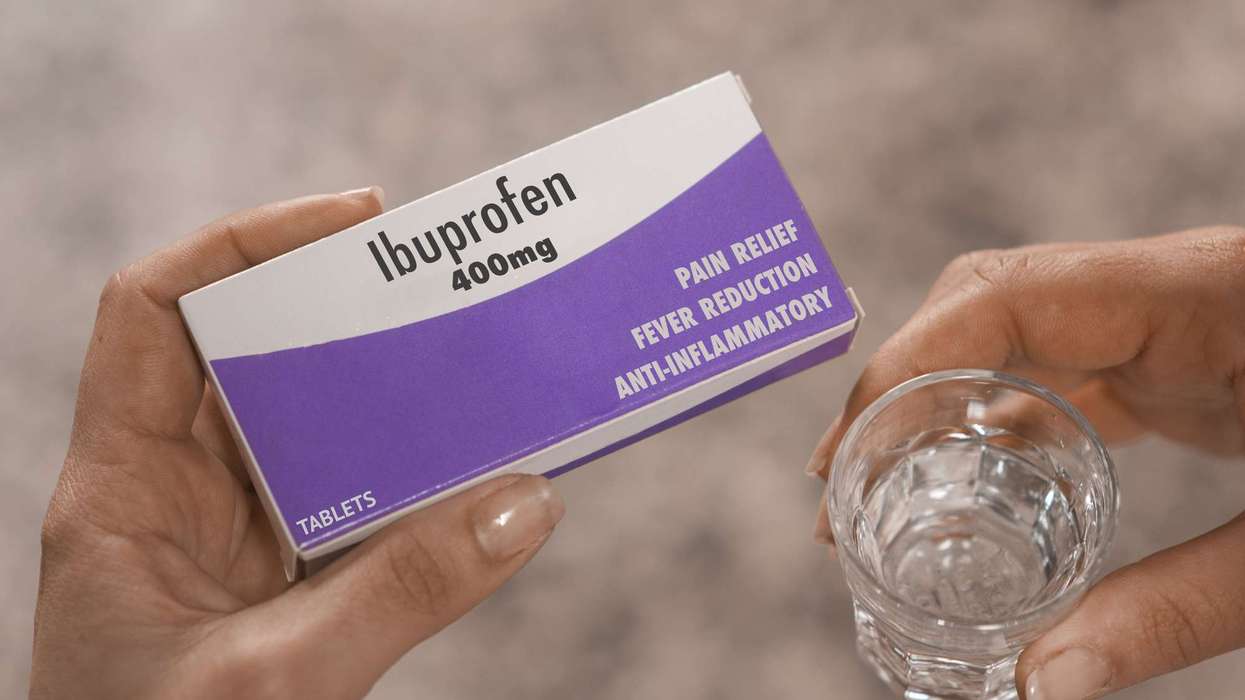
However, deuruxolitinib also has side effects such as headache and acne.
“Deuruxolitinib can only be obtained with a prescription. The recommended dose is an 8 mg tablet to be taken twice a day,” said Julian Beach, MHRA executive director, Healthcare Quality and Access.
He said the MHRA will review the safety and effectiveness of deuruxolitinib.