Key Summary
- Manufacturer alerted MHRA about the wrong Braille text on the outer packaging of the medication.
- Five batches are reported to have the error.
- The medicines will not be recalled as the error does not cause any harm to the patients.
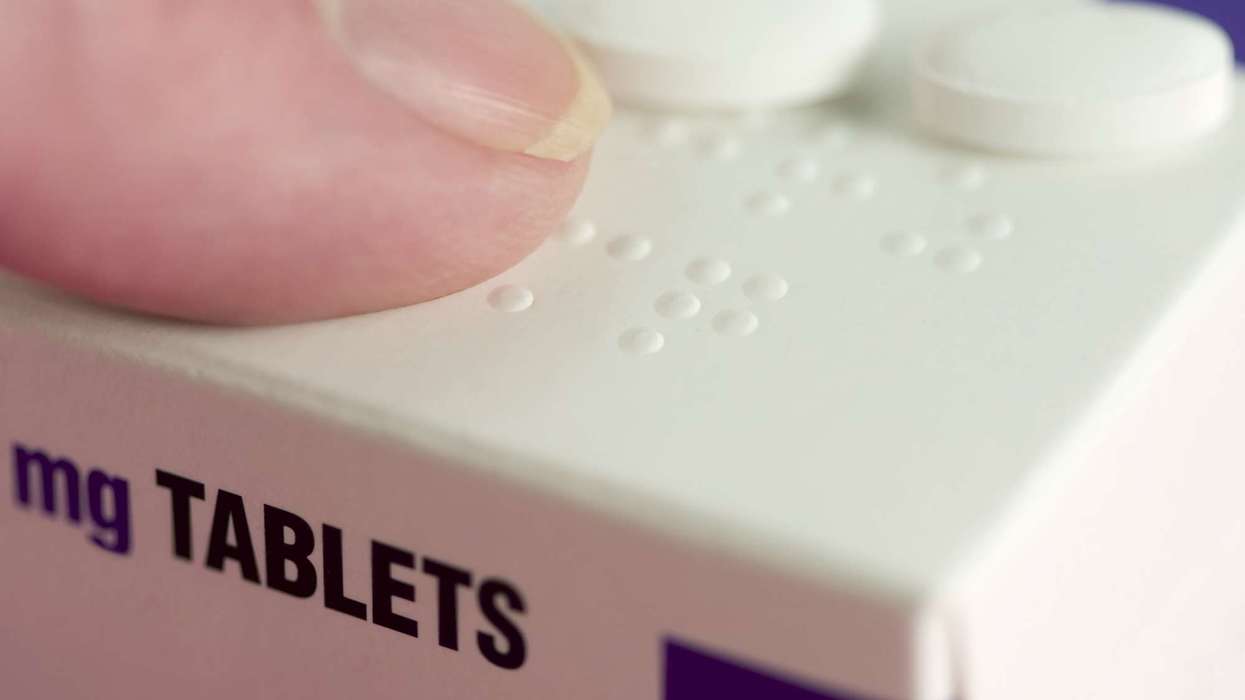
Doncaster Pharma has informed the Medicines and Healthcare products Regulatory Agency (MHRA) that there is an error related to Braille embossing on the packets of some imported batches of Hiprex 1g tablets (POM), for recurrent urinary tract infections (UTIs).
Five batches of Hiprex (251352/BA, 251358/BA, 252137/BA, 252137/BB, and 251358/BB) have been reported with incorrect Braille text ‘1mg’ instead of ‘1g’ on the outer packaging.
However, the printed text on the outer packaging, immediate packaging and Patient Information Leaflet (PIL) is correct.
As there is no problem with the medicine quality, the batches are not recalled.
As the products are supplied in a single strength, there is no risk of dosing errors arising from the Braille issue.
Health professionals will explain the errors to patients who depend on Braille.