Key Summary
- Two deaths in Northern Ireland have been reported to the Medicines and Healthcare products Regulatory Agency and are being reviewed for a possible link to weight-loss drugs.
- The reports are part of over 500 suspected GLP-1 side-effect cases, though the regulator says the deaths may also be linked to other health conditions.
- Research suggests people often regain much of the lost weight after stopping the injections, highlighting the need for long-term lifestyle changes.
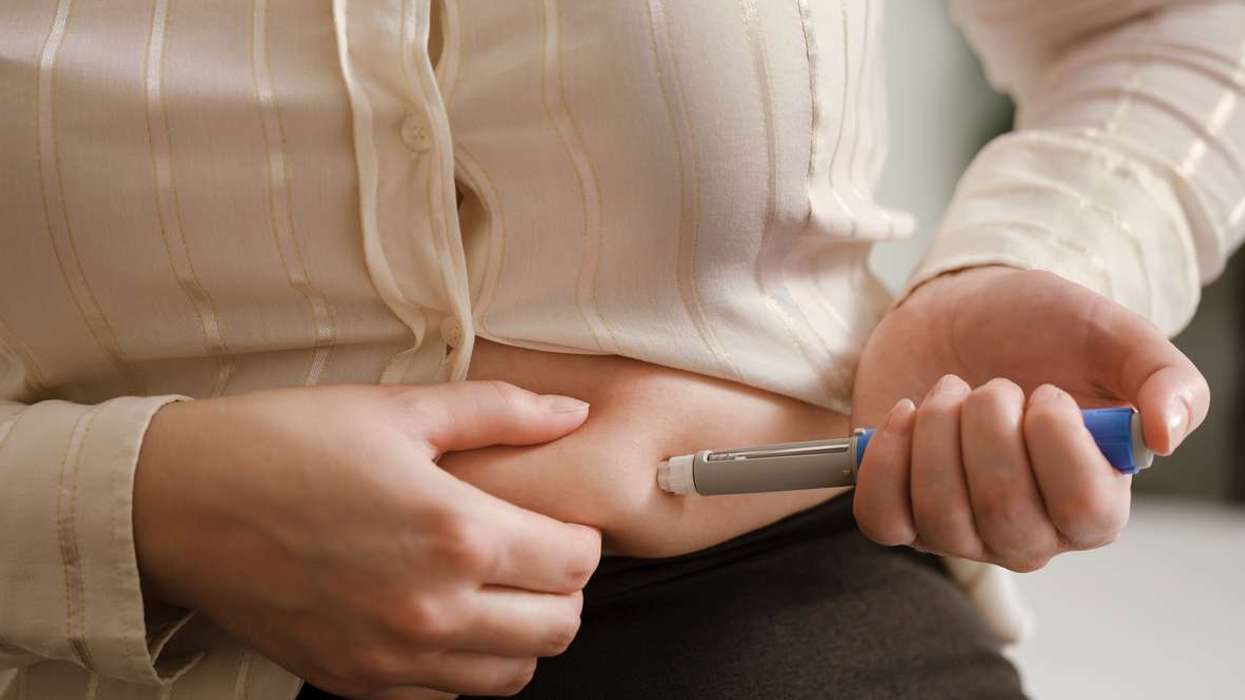
The Medicines and Healthcare products Regulatory Agency (MHRA) has received reports of two deaths from Northern Ireland, potentially caused by weight loss drugs.
A man and woman aged between 40 and 60 years have died in Northern Ireland.
They are among the other 511 reported cases of GLP-1 adverse drug reaction reports from Northern Ireland known to the MHRA between 2024 and 2025.
While one person who died took Mounjaro (active ingredient tirzepatide), the other one used an unspecified semaglutide medication, reported the BBC.
Mounjaro, Wegovy, Ozempic, Rybelsus, etc. are some of the popular weight loss drugs widely used by the public.
As per University College London 1.6 million adults in England, Scotland and Wales have used these weight loss jab during 2024 and 2025 alone.
However, the MHRA has responded saying the current scenario is only suspicion and the cause of deaths could be other reasons like underlying or concurrent illnesses as well.
MHRA had already warned the public regarding the risk of acute pancreatitis as a common side effect of GLP-1 in January.
The MHRA Yellow Card Scheme has 317 cases of adverse reaction from Mounjaro, 135 cases from Wegovy and 12 from Ozempic.
Based on the history of reaction a common form of adverse reaction in Northern Ireland is gastrointestinal disorder with 327 cases during 2024 and 2025.
Nervous system disorders, skin and subcutaneous tissue disorders, psychiatric disorders, musculoskeletal and connective tissue disorders, cardiac disorders, reproductive system and breast disorders, etc. are some other commonly found reactions.
Similarly, a recent study by University of Cambridge involving 3,200 people with six trials, also reveals that stopping weight loss drugs results in rapid weight gain among the patients.
The study shows that patients regain nearly 60 per cent of their lost weight within a year of stopping the drug.
“Drugs such as Ozempic and Wegovy act like brakes on our appetite. When people stop taking them, they are essentially taking their foot off the brake,” commented Brajan Budini, a medical student from School of Clinical Medicine and Trinity College, University of Cambridge.
"If the regained weight is disproportionately fat, individuals may ultimately be worse off than before in their fat-to-lean mass ratio, which may have adverse consequences for their health,” Budini added.
However, the researchers also say that nearly 25 per cent of the initial weight loss sustains in the patients for the long term.
Dr Mitra Dutt, associate medical director at Lloyds Pharmacy Online said, “As a doctor who prescribes weight loss injections in clinical practice, I see first-hand how effective these medications can be, but also the challenges patients face when treatment stops. That’s why I tell patients on day one: if you take your foot off the brake, your appetite will return. Without engrained changes to lifestyle and habits, weight regain is common.”
Following this, experts state that how weight loss drugs cannot be treated as a cure along with how treacherous they could get.
It is important to identify one’s health condition and choose the right weight loss drug to stay healthy.
The MHRA has guaranteed to prioritise patient safety by strictly monitoring GLP-1 drugs via the Yellow Card Scheme and act promptly to resolve these risks.