Key Summary
- The MHRA has authorized donidalorsen (Dawnzera) for patients aged 12+ to prevent attacks of hereditary angioedema (HAE).
- Trials showed a drastic reduction in monthly attacks, with patients on a 4-week dose seeing an average of just 0.4 attacks compared to 2.3 for those on a placebo.
- The drug works by targeting plasma kallikrein to lower bradykinin levels, the primary trigger for HAE swelling.
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved donidalorsen (Dawnzera) injection for people aged 12 and older to prevent hereditary angioedema (HAE) attacks.
The approval was granted to Otsuka Pharmaceutical Netherlands B.V.
Donidalorsen injection is administered from a pre-filled pen under the skin. It blocks plasma kallikrein production, curbing bradykinin buildup in bloodstream that sparks HAE swelling.
In a 24-week trial with 91 participants show promising results, with those administered donidalorsen every 4 weeks showing marked improvement from those who took placebo.
It also boosted quality of life, per validated survey on symptom interference.
The condition
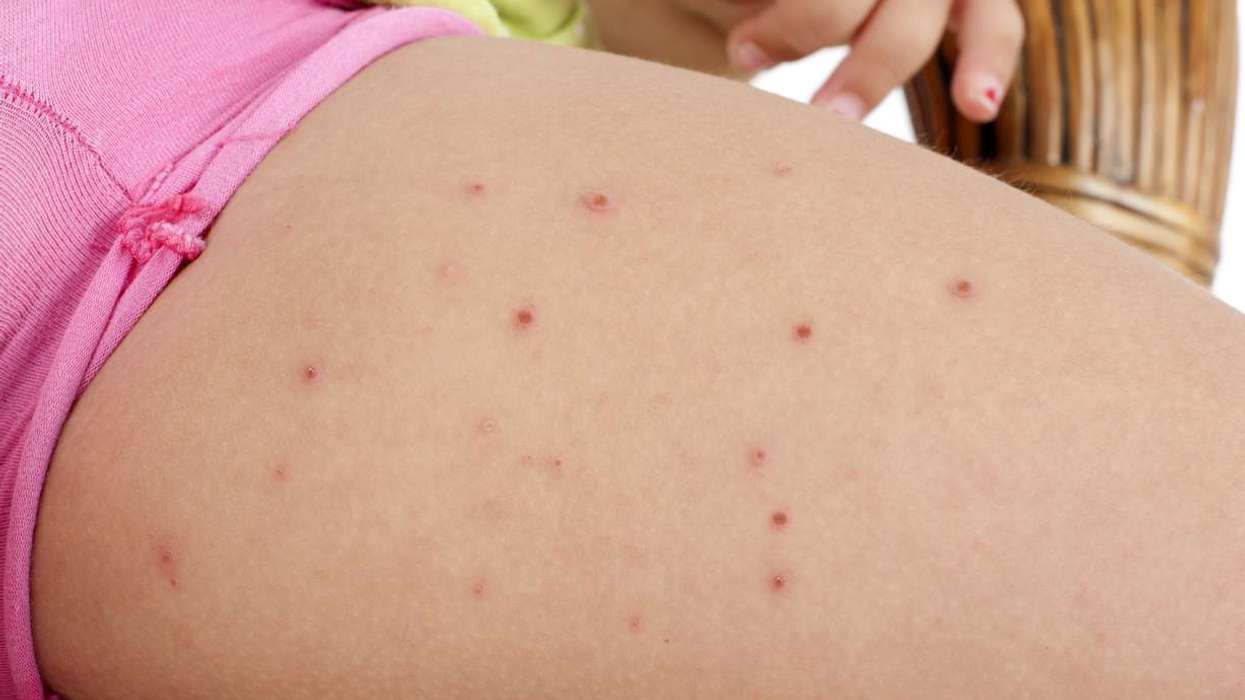
HAE is an inherited condition characterised by a deficiency or dysfunction of the C1 inhibitor protein.
This can lead to swelling around the body and stomach pains. These attacks can be life-threatening when swelling around the throat presses against the airway.
Donidalorsen works by inhibiting the production of plasma kallikrein, thereby reducing bradykinin levels in the bloodstream. This helps to prevent the symptoms associated with HAE.
Julian Beach, MHRA Executive Director of Healthcare Quality and Access, said: “As with all medicines, we will continue to closely monitor the safety and effectiveness of donidalorsen as it is used more widely.”
Common side effects (affecting over 1 in 10) involve injection-site reactions such as redness, discoloration, pain, itching, hardening, bruising, scaling, hematomas, allergies, or swelling. Liver enzyme shifts may appear in bloodwork.