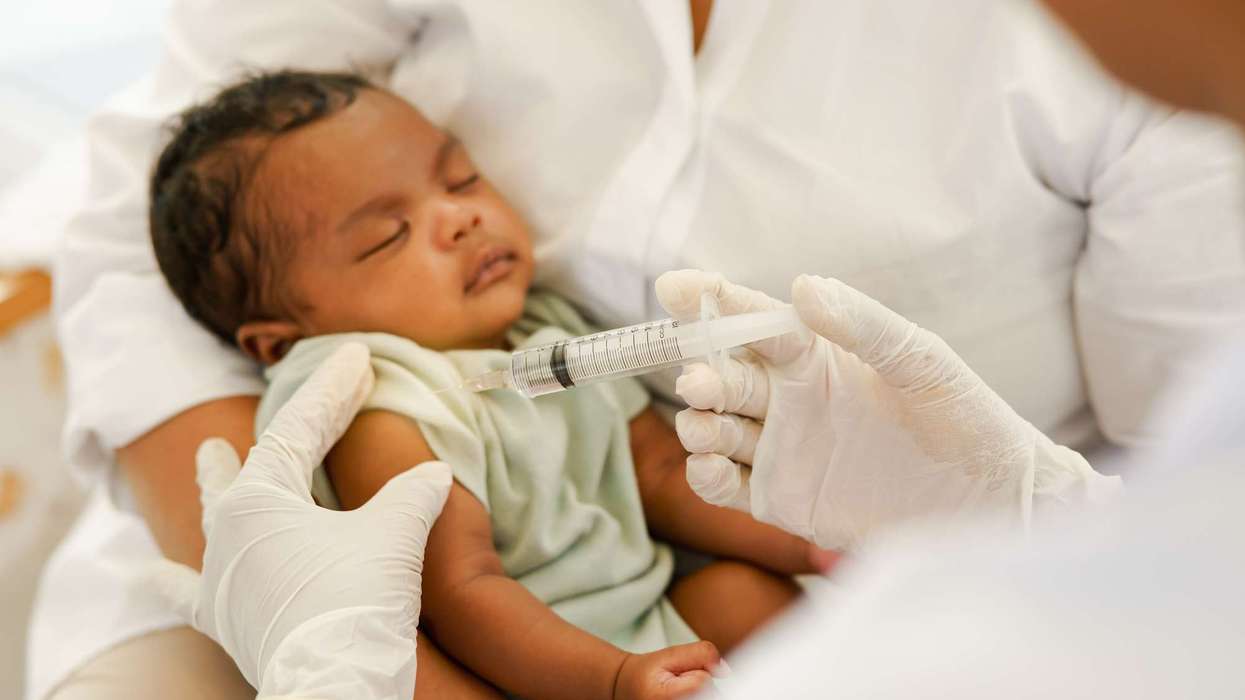
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved Enflonsia (clesrovimab-cfor) injection, to safeguard newborn infants from respiratory syncytial virus (RSV), a lower respiratory tract illness.
Clesrovimab, the active ingredient in the drug is a powerful antibody that fights the harmful virus, and it is manufactured by Merck Sharp Dohme (UK).
It is a single jab to be administered by a health professional, and infants up to 12 months of age are eligible.
RSV is a seasonal and contagious disease that resembles common cold, which can lead to severe lung disorders such as pneumonia, infant bronchitis, if left unattended.
So, the health experts urge the parents to vaccinate their children against it before they turn two.
“It’s important that we do our best to protect our newborn babies and infants especially throughout RSV season,” commented Julian Beach, MHRA executive director of Healthcare Quality and Access.
He added, “Part of this is enabling access to high quality, safe and effective medical products are key priorities for us.”
Although approved, MHRA guarantees to constantly review the medication etc. to ensure public safety.
In case of any side effects such as pain, swelling, redness or rashes at the injection site, people are urged to seek immediate medical help and report the incident to the MHRA Yellow Card Scheme.