Key Summary
- Milpharm Ltd issued a defect alert for two batches of Loperamide hydrochloride tablets over leaflet errors.
- The PIL gives incorrect instructions and misses a rare side effect.
- Patients can still use the medicine, but pharmacists should explain the correct use.
Milpharm Limited has issued a Class 4 medicine defect notification, due to discrepancies in Patient Information Leaflet (PIL) for two batches of Loperamide hydrochloride 2mg Orodispersible Tablets.
The medication is used to treat short-term diarrhoea in adults and children over 12 years.
It is also used to treat irritable bowel syndrome (IBS) diarrhoea in adults over 18 years.
The notification for 12 pack pills with batch number 25882X3 and the 6 pack pills with batch number 25882X2, containing the active ingredient Loperamide hydrochloride, was issued on 11 May.
The company identified two errors on the medication’s PIL.
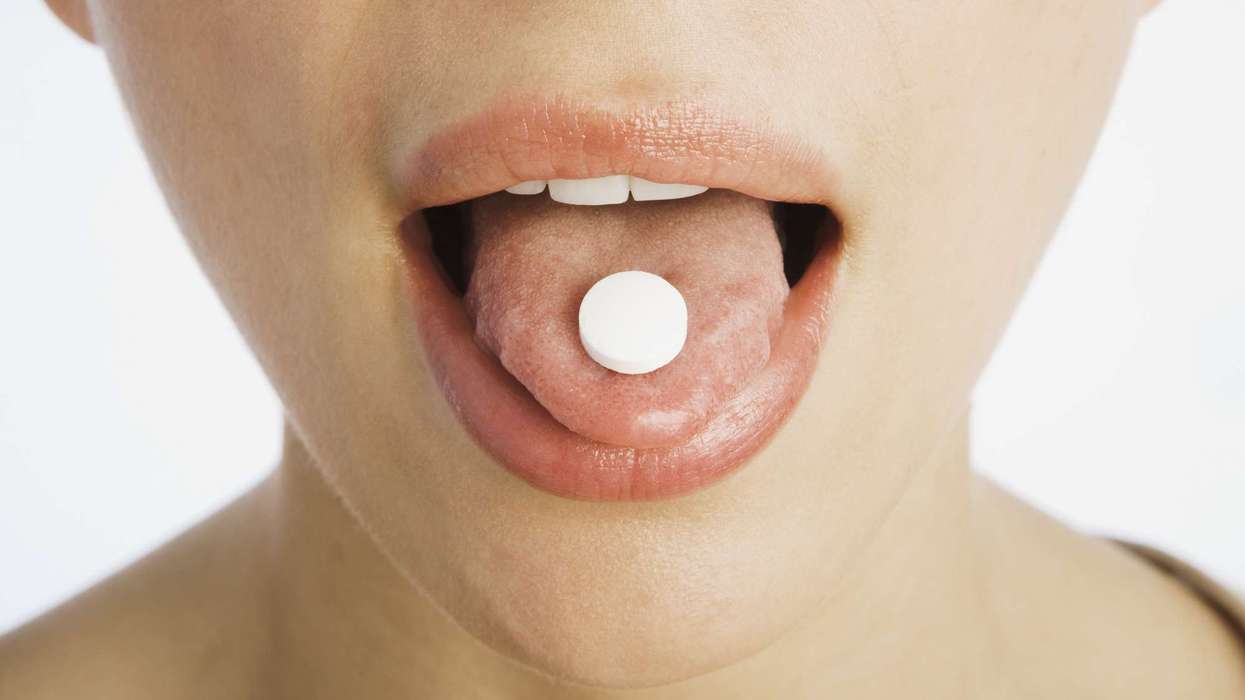
Under Section 3, Method of administration, the PIL of both the packages wrongly advises the patients to swallow the orodispersible tablet fully with water.
In reality, the tablet must be placed on the patients’ tongue allowing it to dissolve without the help of water.
Along with that, under Section 4, Rare side effects, the PIL also has omitted “Burning or prickling sensation of the tongue,” (seen in 1 among 1,000 patients), which is a rare yet, unskippable side effect of the orodispersible formulation that is listed in the Imodium Instants reference PIL.
However, Milpharm can confirmed that all the other PILs carry all the necessary information such as indication, dose, dosing frequency, maximum daily dose, etc., without fail.
So, the healthcare professionals are advised to note the batch numbers and share the information, especially how to consume and the side effects, with the patients as required.
The right instructions are printed on the drugs’ carton.
The professionals can also assure the patients that there is no need to return the medications as there is no issues regarding its quality.
Patients who face any form of adverse reaction can report it to the MHRA Yellow Card Scheme.