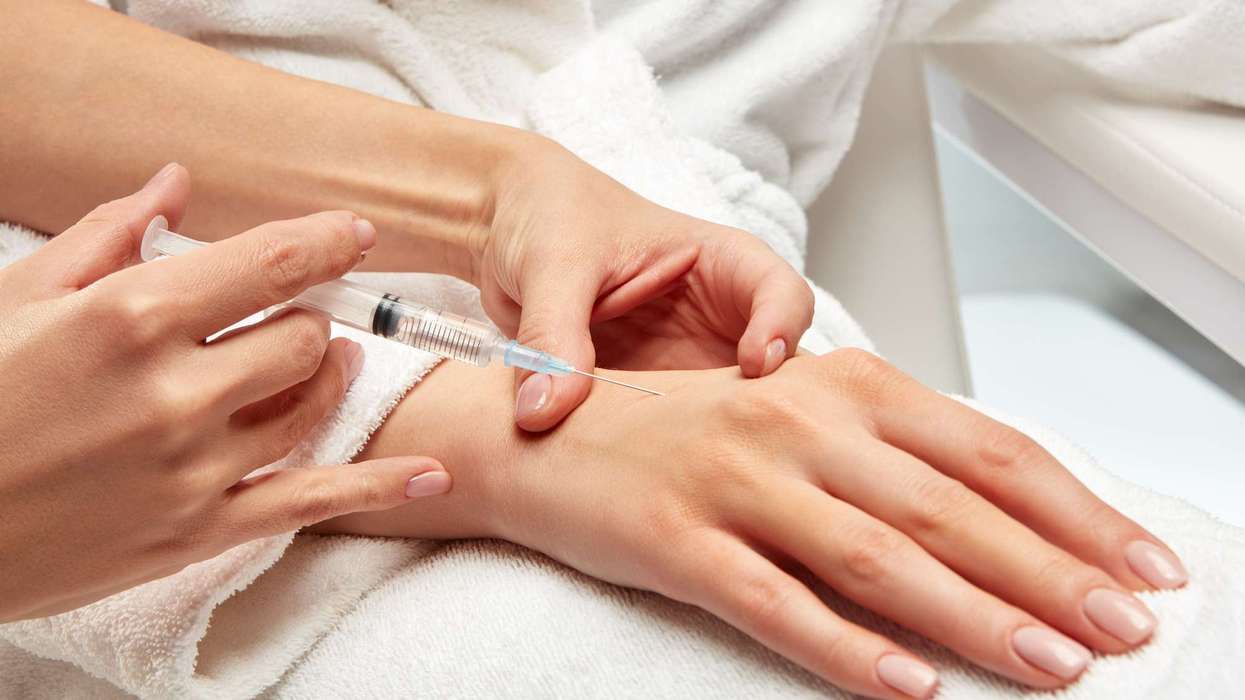
National Institute for Health and Care Excellence (NICE) today recommended Teva’s migraine drug fremanezumab, sold under the brand name Ajovy, for NHS use.
The final draft guidance published by the agency recommends the drug for preventing chronic migraine in adults where at least three previous preventive treatments have failed.
The agency said the decision would pave way for up to 10,000 people to receive the drug on the NHS in England and Wales.
“We are pleased that the company has been able to work with us to address the concerns highlighted in the previous draft guidance so that we are now able to recommend fremanezumab as an option for people with chronic migraine when several other medications have failed,” said Meindert Boysen, director of the Centre for Health Technology Evaluation at NICE.
Teva will provide the drug, which costs around £5000 a year, at a discounted price after agreeing for a confidential commercial arrangement.
NICE said the improved discount makes it a “cost-effective” use of the NHS resources.
The recommendation also includes people with chronic migraine for whom botulinum toxin type A has failed.
“Teva is very pleased to receive this decision by NICE as we seek to expand the availability of AJOVY in the UK in an effort to ensure that patients living with migraine have access to this treatment option,” said Kim Innes, general manager of Teva UK and Ireland.
“We’re committed to improving the lives of migraine patients, and believe that the introduction of AJOVY, having been designed specifically for migraine prevention and with its flexible dosing options, will bring new opportunities and improve the lives of patients struggling to control this disabling condition.”
It is estimated that there are 190,000 migraine attacks experienced every day in England with women more likely to experience one than men.
The drug offers both quarterly and monthly dosing options for the prophylactic treatment of migraine, with the flexibility to be self-injected at home by the patient (or carer).
“Anyone who looks after people with chronic migraine understands just how debilitating this neurological disorder can be,” commented Dr Mark Weatherall, president of the British Association for the Study of Headache.
"We have waited a long time for this new class of drug to be made available in the NHS, but now that we can prescribe fremanezumab, I am excited to see what a difference it will make to the lives of many of my worst affected patients."
Fremanezumab belongs to a class of treatments called anti-CGRP (calcitonin gene-related peptide) monoclonal antibodies, which have been specifically designed to target the underlying causes of migraine. It is first anti-CGRP drug recommended by NICE.
The drug is also approved in Scotland by the Scottish Medicines Consortium.
NICE is expected to publish the final guidance next month if no appeals are made by registered consultees.