Concessionary prices for medicines are often treated as shorthand for shortages. If pharmacists are requesting concessions, the assumption goes, they must be struggling to source the products they need. Concessionary prices are better defined as a financial pressure-release valve when products cannot be purchased at or below the Drug Tariff reimbursement price.
It sounds intuitive that a concessionary price equals a shortage - but it is increasingly misleading, and risks obscuring the real pressures facing the whole medicines supply chain. In fact, one of the great strengths of the UK pricing and reimbursement system is that, for the vast majority of the time, generic prices are very low - among the lowest in Europe.
However, in times of constrained supply or rising costs, manufacturers can raise prices to attract stock to the UK rather than to other global destinations. This often manifests itself in concessionary prices.
Indeed, from a manufacturing perspective, the correlation between concessionary prices and shortages is far too superficial to be meaningful. The data tells a more nuanced story.
For the past five years, Medicines UK has analysed NHS data monthly to track the number of patented and off-patent medicines experiencing supply issues.
Throughout this period, shortages have followed a clear pattern: more than 100 products a month at their peak from September 2023 over the next 12 months, falling to around 60 today - the lowest level in more than three years.
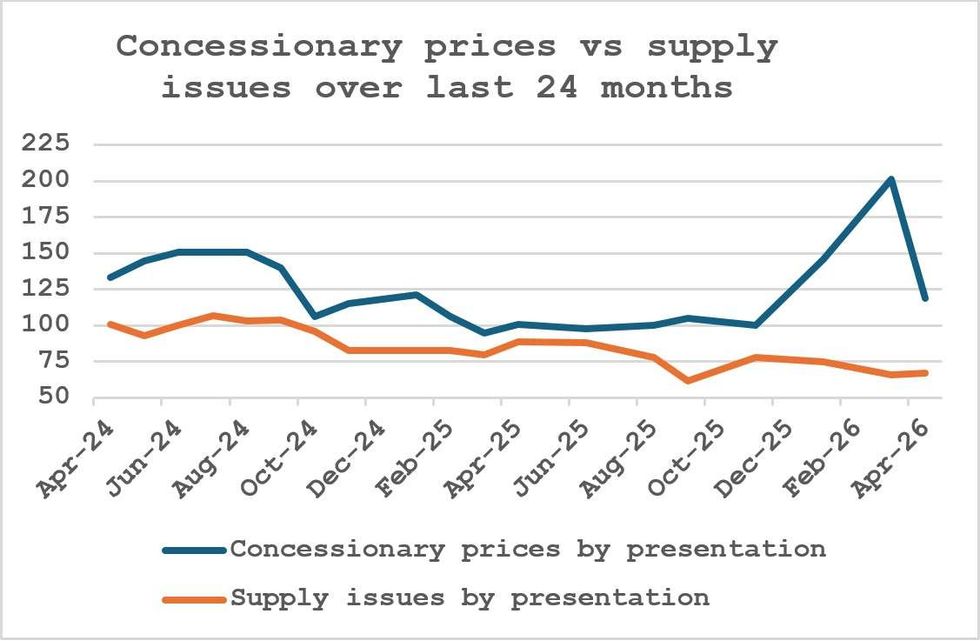
Yet over the same period, the number of concessionary prices has fluctuated dramatically, often diverging sharply from actual supply issues. In some months, concessions have risen steeply even as shortages have plateaued or fallen. The start of this year is a striking example: a noticeable rise in concessions despite stable supply.
This divergence matters. It challenges the prevailing narrative that concessions automatically signal a lack of stock. In reality, they increasingly reflect a reimbursement system struggling to keep pace with market conditions or the growing use of algorithmic procurement.
The overlaid graphs below, which track concessions and supply issues, show a clear and persistent gap. And that gap has consequences. Too much of the pharmacy margin is absorbed by concessionary-priced products, diverting funding away from other products in Category M - medicines prescribed by their generic names that are commonly available.
With only a finite amount of margin available, this then makes it less likely that the reimbursement tariff for other Category M medicines will be enough in subsequent months. The margin is being stretched too thinly to sustain a well‑functioning system.
The reimbursement framework itself is creaking. While the system is still fit for purpose and adept at delivering taxpayer value for money and providing a release valve to enable supply problems to be addressed as efficiently as possible, it has not undergone meaningful reform for more than a decade, despite the pressures facing community pharmacy. The number of packs dispensed has grown by 15 percent over that period, while operating costs have risen by nearly 40 percent due to inflation. This trajectory is simply unsustainable.
At the same time, community pharmacists are being asked to make better use of their clinical expertise. While this supports the 10-year plan to ease pressure on other parts of the health system and promote more preventive care, it adds extra demands on community pharmacy.
Greater overall funding through a revised Community Pharmacy Agreement is clearly needed. Negotiations continue, and time will tell what is concluded. But better, more timely data could also make a significant difference.
At present, manufacturers and wholesalers provide actual selling price data to the Department of Health & Social Care every three months. This data inputs into reimbursement calculations. But the data may be out of date by the time it is used to inform prices. It lags behind and fails to reflect current market dynamics.
This government-administrative lag in the reimbursement system means that Drug Tariff prices are always lagging behind market dynamics. A closer-to-real-time system could also help reduce the scale of government discount enquiries in which pharmacies have a deduction applied to their payment to correct a previous overpayment.
We are looking at whether more frequent reporting would help spread margin more equitably and promote greater predictability and less volatility across the market – provided this reporting does not place an unsustainable burden on manufacturers.
This reporting, in turn, could support a more resilient and sustainable supply chain for patients, pharmacies, and the NHS.
The evidence is clear: concessionary prices are not a reliable indicator of shortages, but a warning sign. The basis of the pricing and reimbursement system works well. It delivers taxpayer value and security of supply, but the model is being stretched to its limits.
With better data, fairer margins and a funding model that reflects the true cost of dispensing, we can restore stability to the supply chain and protect patient access. The choice now is whether to act with foresight - or wait for the system to further fracture under the weight of avoidable pressure.
(Mark Samuels is the chief executive of Medicines UK)