Key Summary
- Medicines and Healthcare products Regulatory Agency approves 7.2mg single-dose semaglutide (Wegovy) pen for adults with obesity
- Max weekly dose (7.2mg); only for eligible patients (BMI ≥30) under clinician guidance
- Safety monitoring continues; side effects must be reported via the MHRA Yellow Card Scheme
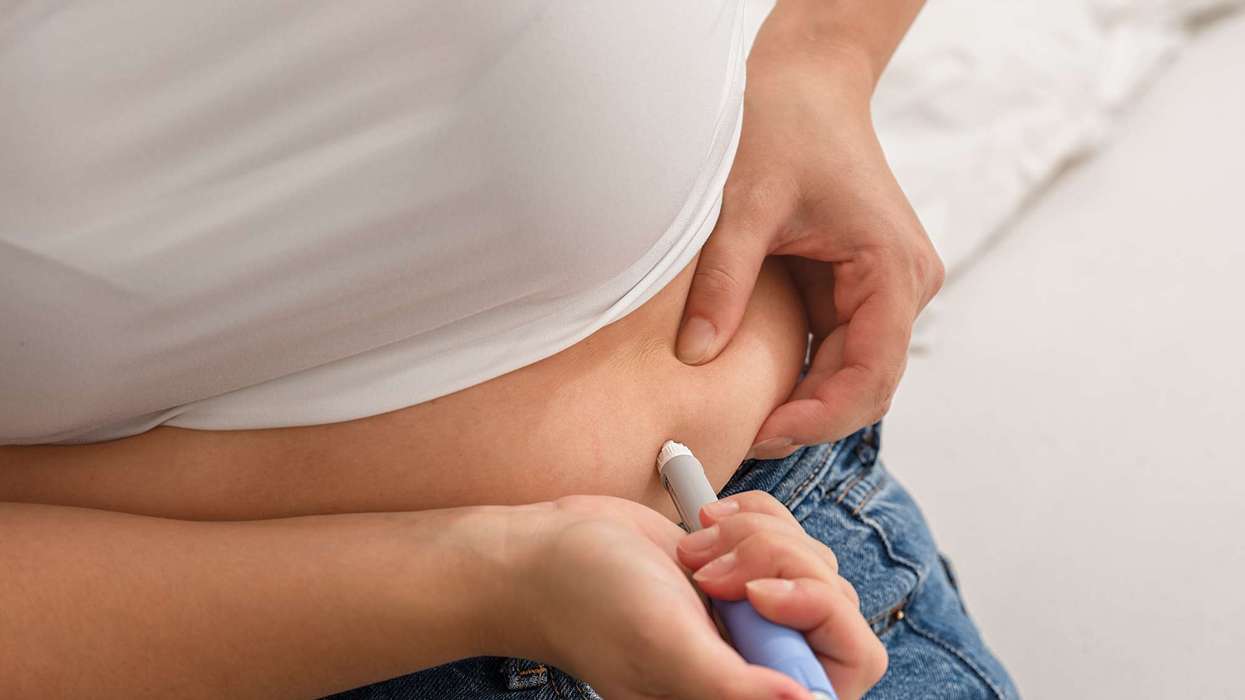
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved the new single-dose 7.2mg semaglutide (Wegovy) pen for treating adult obesity.
The 7.2mg dosage of the drug is given through one injection.
As the starting dosage of Wegovy is 0.25mg per week, it could eventually rise every four weeks if needed.
However, 7.2mg is the maximum dose per week.
The treatment is accessible to patients with a Body Mass Index (BMI) of 30kg/m² or higher, which helps them receive the maximum weekly dose for weight loss and weight management.
The MHRA has strictly advised that people with lower BMI or people who take Wegovy to reduce heart health risks must not administer this dose.
Eligible patients must only use the drug based on the guidance of their prescriber, doctor, pharmacist or nurse.
This decision follows the January 2026 authorisation of the 7.2mg maximum weekly dose, which required three 2.4mg doses administered on the same day using the standard 2.4mg pen.
Although approved, the MHRA will keep reviewing the drug to ensure public safety.
Any form of its adverse effect must be reported via the MHRA Yellow Card Scheme.