Key Summary
- The patent for semaglutide will expire in 10 countries this year, but the UK patent will remain till 2031.
- Generic drug makers will not be able to export to UK, and this will give rise to smuggling as the demand is high.
- Without professional guidance, there will be no support for managing side effects, which are common and sometimes dangerous.
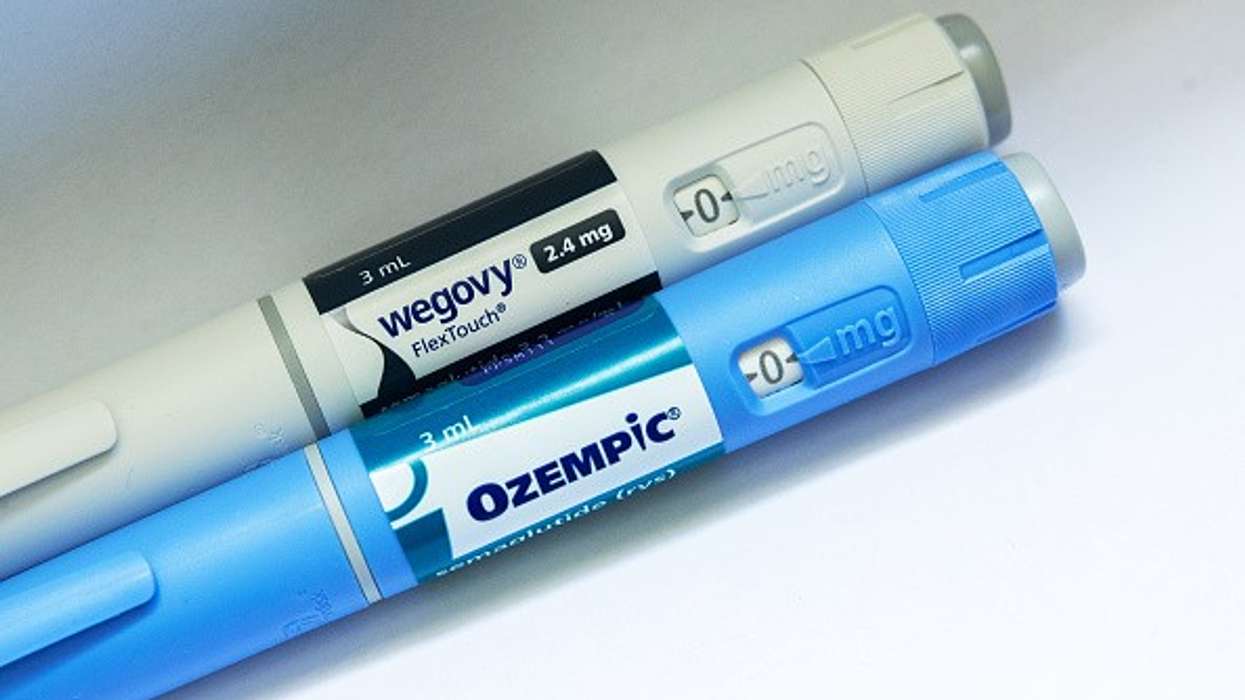
The patent for semaglutide, the active ingredient in Novo Nordisk's weight-loss drugs Wegovy and Ozempic, expired in India in March, and by the end of 2026, it will expire in 10 countries, including Brazil, China, South Africa, Turkey, and Canada.
With India, Brazil and China being global powerhouses of generic drugs, it is expected to trigger a wave of generic versions of Wegovy and Ozempic, and analysts expect the prices to go down by 50 percent.
Semaglutide belongs to a class of medicines known as GLP-1 receptor agonists, which mimic a hormone that regulates appetite and blood sugar.
Originally developed to treat diabetes, these drugs are now seen as game changers in weight loss treatments.
In India, many major companies such as Sun Pharmaceutical Industries, Dr Reddy's Laboratories, Zydus Lifesciences, Lupin, and Mankind Pharma rolled out their semaglutide versions on March 21, a day after the patent expired. By the end of this year analysts expect around 50 brands from India itself.
However, in the US, UK, and many parts of Europe, the patent protection for semaglutide would last till 2031 and beyond, as they have patent term extension mechanisms that can be used to extend the protection period.
This means that the generic makers of semaglutide will not be able to export their products to these countries for another five years.
MedExpress head of Medical Affairs Dr Sophie Dix told Pharmacy Business that this will widen the price gap between accessing semaglutide through regulated clinical pathways in the UK and generics available in international markets.
"This disparity is the primary engine for the grey market.
"Countries such as Brazil and China are major pharmaceutical manufacturing hubs that will be able to quickly scale production of generic medications. "
Unregulated pathways
She pointed out that generic drugs are different from counterfeit medications (such as retatrutide) as they are manufactured legally.
"It is the cross-border supply that is unregulated. They will likely follow existing illegitimate supply routes already used for Prescription-Only Medicines (POMs) such as sleeping tablets, nootropics and pain killers."
MedExpress had recently surveyed 2,000 UK adults who are using, have previously used, or are considering weight loss medication.
They found that 64 percent have already taken action in response to price changes, and many of them admitted they would consider unverified sources following price increases.
"The danger isn't the existence of generics, but the supply of illegal ones. The risk is the complete absence of lack of clinical support for patients and concerns about the supply chain.
"It also creates yet more confusion for patients."
No clinical insight
The most immediate risk of using unlicenced or imported weight-loss products is the complete absence of clinical oversight.
"Without a healthcare professional to manage dose titration and monitor for side effects, people are essentially self-medicating with a powerful treatment.
"In a regulated environment, trusted medical experts provide ongoing support to ensure medication is working safely. Without that, patients are left to navigate complex dosing (or titration), side effects and potential adverse reactions entirely alone," Dix said.
Lloyds Pharmacy Online Doctor chief medical officer Dr Kieran Seyan, also concurred with this view.
He told Pharmacy Business, “Medications like these are not one-size-fits-all treatments and require clinical oversight, including careful patient selection, dose titration and ongoing monitoring, to ensure they are used safely and effectively.
"Where there is limited access or affordability constraints, there is a risk that patients may turn to illicit or unregulated sources. This can pose significant risks, particularly where there are no guarantees around product quality, appropriate dosing, or follow-up care.
“The priority should be ensuring patients can access these treatments through trusted, regulated providers. This ensures not only that the medication itself is safe and appropriate, but that patients receive the wider clinical support needed to achieve sustainable, long-term outcomes.”
Dangers to patients
Dix also listed out the other major dangers these medicines could pose to the public.
Lack of Consultation: By bypassing essential clinical consultations, a medical professional cannot ensure the medication is suitable for the individual, and GLP-1s are not suitable for everyone. Without professional guidance, there is no support for managing side effects, which are common and sometimes dangerous.
Dose Accuracy: GLP-1s require precise titration. If a generic is supplied by vials, it will lack the calibrated delivery systems of licensed pens, risking accidental overdosing.
Cold Chain Failure: Semaglutide is temperature-sensitive. Illicit shipping rarely maintains the strict 'cold chain' required, meaning patients may be injecting degraded, ineffective, or biologically unstable proteins.
"The unregulated 'grey market' also creates a dangerous transparency gap; once you step outside of a regulated pharmacy, it becomes nearly impossible for a consumer to distinguish a legitimate unlicensed import from a dangerous counterfeit produced in a clandestine lab," she said.
Cosmetic use
Dix also expressed concern over the shift in use of weight-loss drugs from a medical necessity to 'cosmetic' use, especially by those aspiring to have a 'perfect' body shape.
Citing MedExpress data, she said that over half (54 percent) of people first hear about GLP-1s through social media, while 30 percent learn about them from influencers.
"When these powerful metabolic hormones are marketed as 'lifestyle' products - fueled by social media and cheap, unregulated generics - the perception of risk is dangerously lowered.
"GLP-1 medications are not magic erasers for a 'perfect body', but rather act as a powerful catalyst for profound health transformation.
"When used correctly, GLP-1s are a medical intervention that helps reset a relationship with food by working on the brain’s hunger signals and gastric function."
A clinical journey
Dix described weight loss as a "clinical journey, not a transaction."
"The grey market provides a vial, but it doesn't provide a doctor."
She said that weight loss is a journey that works best when supported by professional guidance.
"Without 'wraparound care', patients may find it difficult to manage common side effects or maintain the lifestyle changes needed for long-term success.
"Because GLP-1s are so effective at suppressing appetite, clinical support is vital to ensure individuals are meeting their nutritional needs, particularly around protein intake and activity levels. This helps preserve muscle mass and ensures that weight loss is both healthy and sustainable, rather than just a short-term drop on the scales."
Safe approach
"The safest approach is to always speak directly to a qualified healthcare professional and access through regulated suppliers selling licensed products with clinical support," she said.
In order to check whether the service is genuine, the patients should check the following.
Check for accreditation: A genuine UK online pharmacy must display its General Pharmaceutical Council (GPhC) registration number. You can and should verify this number on the official GPhC website.
Look for clinical regulation: For organisations that provide clinically-led services, check for Care Quality Commission (CQC) registration, which governs the quality of medical care provided.
Question the source: If you are being offered a weight-loss jab via a social media DM, a messaging app like Telegram, or a beauty salon, it is almost certainly illegal. If it seems too good (or too cheap) to be true, it likely is.
Burden on NHS
A major burden of people seeking treatment from dubious sources comes as an added burden for the NHS in the form of corrective or remedial treatments.
"The irony of the grey market is that trying to save money privately often results in a massive cost to the taxpayer. The NHS is already under immense strain and does not have the capacity to act as the 'cleanup crew' for unregulated medicine," Dix said.
"With unregulated products entering the market, there could be a rise in hospital admissions for acute complications, such as severe dehydration, electrolyte imbalances, or acute pancreatitis caused by unmonitored or incorrect dosing.
"Patients may delay seeking help for side effects because they may be reluctant to admit to their GP that they have been taking unregulated medication. This could lead to situations where patients may also misattribute symptoms to their illicit GLP-1 medication, potentially missing the early diagnosis and treatment of unrelated illnesses, and as a result, poorer health outcomes for patients and increased costs to the NHS.
"Looking forward, there could also be potential for long-term remediation, addressing the metabolic fallout and nutritional deficiencies in patients who have used these drugs improperly for years without monitoring.
"The most cost-effective and health-effective route is the direct one. By bypassing unregulated sources, patients protect their own health and preserve the resources of our national health service," Dix said.